The purpose of this experiment was to use P-anisaldehyde to synthesize 3,5 disubstituted isoxazoline in a cost-effective way while producing a reasonable yield. Finding a cost-effective way to synthesize the compound in large quantities with reasonably high yield will lower the production cost for medicines with numerous applications. The final product has many useful purposes including: being a precursor to a compound that can combat Type I diabetes, an antagonist of Macrophage Migration Inhibitory Factor (MIF), and may be a new target used to counter Type II diabetes (Mosher & Norman), (Maurya, Gupta, & Ahmad) (Sánchez-Zamora, Yuriko I., & Miriam Rodriguez-Sosa).
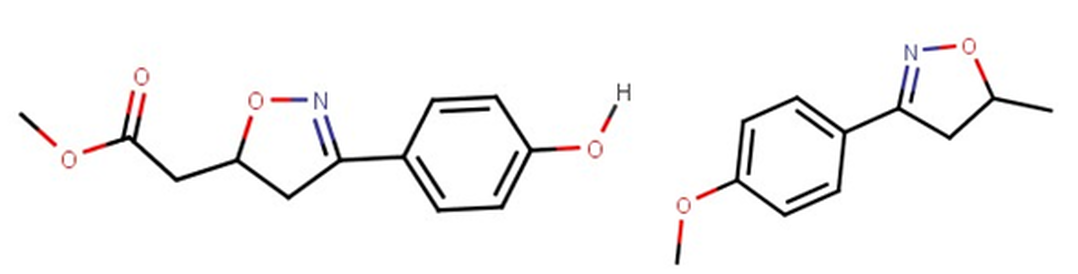
Figure 1: The above compound on the left is called ISO-1, which is structurally similar to the isoxazoline produced by the research on the right. Because of the similar structure, both compounds should produce the same effect.
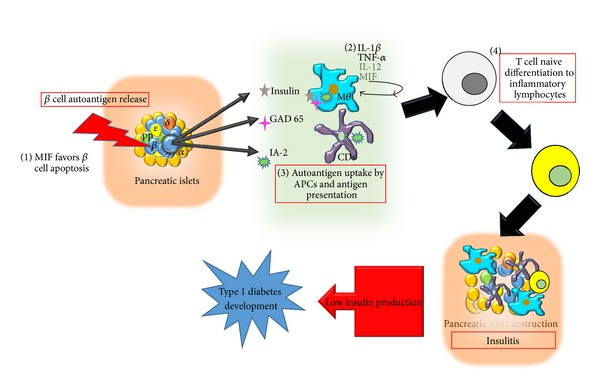
ISO-1 inhibits Macrophage Migratory Inhibition Factor (MIF) which plays a role in both types of diabetes (Mosher & Norman) (Sánchez-Zamora, Yuriko I., & Miriam Rodriguez-Sosa). MIF is a pro-inflammatory cytokine which acts as an alarm bell to the immune system (Sánchez-Zamora, Yuriko I., & Miriam Rodriguez-Sosa). Type I Diabetes is caused by the destruction of Beta cells in the pancreas, mainly by the immune system. MIF is directly involved in Type I Diabetes as it promotes the apoptosis of Beta cells in the pancreas by signaling the immune system to destroy the Beta cells in the pancreas (Sánchez-Zamora, Yuriko I., & Miriam Rodriguez-Sosa). Shown below is the pathway in which MIF activates the immune response (Sánchez-Zamora, Yuriko I., & Miriam Rodriguez-Sosa). By inhibiting MIF through ISO-1, the onset of Type I Diabetes would be slowed. There is a correlation between MIF and Type II Diabetes, but the exact nature of MIF is not known with respect to Type II Diabetes.
Furthermore, another application is that isoxazoline is similar in structure to inhibitors of protein tyrosine phosphatases (PTPase). “A number of these PTPase that act as negative regulators of the insulin signaling cascade have been identified as novel targets for the therapeutic enhancement of insulin action in insulin-resistant disease states like Type II Diabetes” (Maurya, Gupta, & Ahmad).
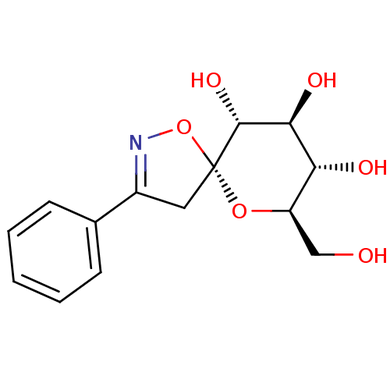
Another form of isoxazoline is a glucose-derived spiro-isoxazoline. This has practical applications in counteracting Type II Diabetes by opposing an enzyme called Glycogen Phosphorylase. It binds to an allosteric site on the enzyme, changing the function of the enzyme by changing its structure (Goyard et al). Glycogen Phosphorylase works by breaking down glycogen into glucose, raising blood sugar levels (Goyard et al). Type II Diabetes is characterized by the target cells not absorbing glucose from the blood stream, leading to high blood sugar levels (Ross et al). This is also known as insulin resistance (Ross et al). By inhibiting an enzyme that releases glucose into the blood stream, there will be less sugar in the blood stream, and it will lower the harmful symptoms of Type II Diabetes.
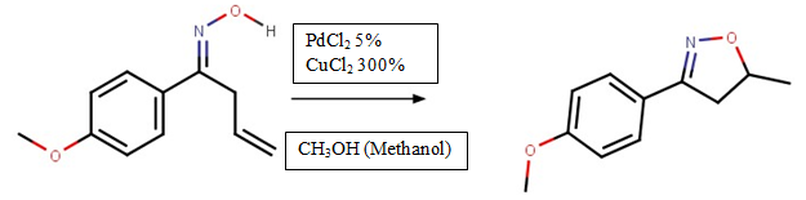
Furthermore, an important reaction in the research is cyclization, which is the formation of a ring shape of atoms instead of a linear structure, which was used to create the isoxazoline in the final reaction. The cost-effective pathway to synthesize the isoxazoline through cyclization will rest mainly on the discovery of a new catalyst that is less expensive than the current catalyst, Palladium (II) Chloride, which cyclizes the oxime into the isoxazoline.
There have been other ways of forming isoxazoline, most notably 1,3 Dipolar Cycloaddition. However, these reactions can be expensive and require complicated procedures. The metal mediated cyclization coupling reaction, using Palladium (II) Chloride or Nickel (II) Chloride, or PdCl2 and NiCl2, is a relatively recent way of producing isoxazoline that is simpler and uses cost-effective starting materials.
There have been other ways of forming isoxazoline, most notably 1,3 Dipolar Cycloaddition. However, these reactions can be expensive and require complicated procedures. The metal mediated cyclization coupling reaction, using Palladium (II) Chloride or Nickel (II) Chloride, or PdCl2 and NiCl2, is a relatively recent way of producing isoxazoline that is simpler and uses cost-effective starting materials.
The main focus of this research is the discovery of a new catalyst to the reaction rather than the expensive Palladium (II) Chloride which sells for more than $900 an ounce. It was hypothesized that Nickel (II) Chloride and Lead (II) Acetate would cyclize the oxime into an isoxazoline because Nickel has similar properties to Palladium and Lead has properties similar to Mercury, which has also been used.