Summary of Reactions:
The procedure consists of 4 main reactions:
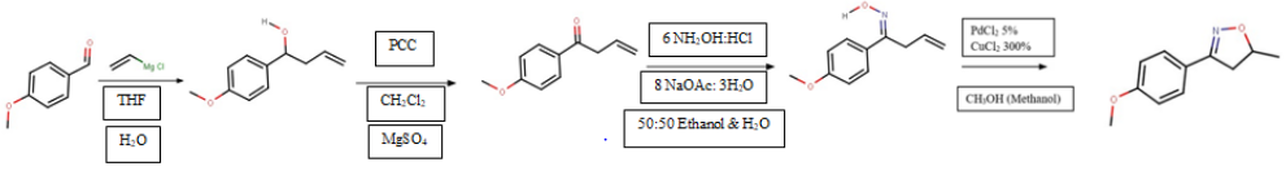
- Grignard Reaction: Changes P.Anisaldehyde into 1-(4-Methoxyphenyl)-3-buten-1-ol
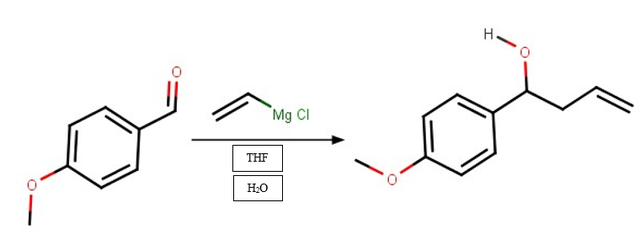
- Oxidation Reaction: Changes 1-(4-Methoxyphenyl)-3-buten-1-ol into a ketone
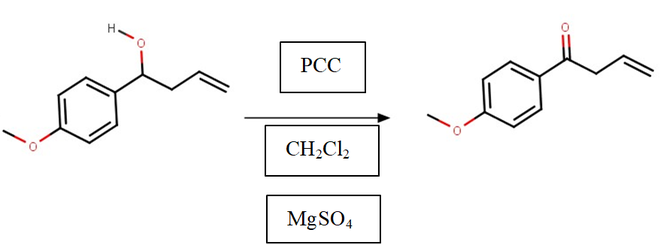
- Oximation Reaction: Changes the ketone into an oxime
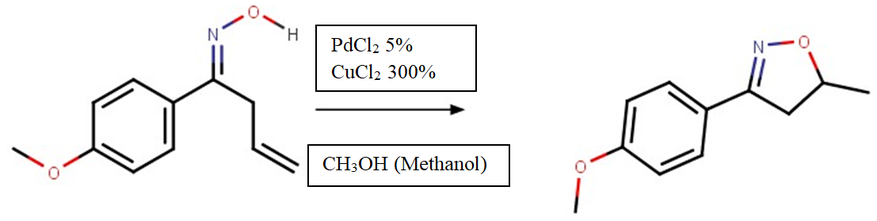
- Cyclization Reaction: Changes the oxime into 3,5-disubstituted Δ2-isoxazoline
1. Grignard Reaction:
The Grignard Reaction lengthened the carbon chain of the starting material, P-anisaldehyde, as well as converting it into an alcohol. This alcohol is called 1-(4-methoxyphenyl)-3-buten-1-ol
2. Oxidation Reaction:
The Oxidation Reaction adds another bond to the oxygen and transforms the alcohol into a ketone.
3. Oximation Reaction:
The ketone is turned into an oxime in this reaction by adding a Nitrogen atom, and the formation of the isoxazole ring is becoming apparent.
4. Cyclization Reaction:
The main focus of our research centers on finding a new catalyst for the cyclization reaction which closes the isoxazole ring. Nickel, Palladium, and Lead were all experimented with, with Palladium as the control.